Information about the Sweedler Research Group.
 In the Sweedler Research Group, we work on a broad array of projects that span analytical chemistry and chemical biology to neuroscience and bioengineering. We conduct research in bioanalytical chemistry and focus on the development of analytical methods for assaying complex microenvironments, including capillary electrophoresis separations, laser-based detectors, MALDI sampling techniques, and nanoliter volume NMR.
In the Sweedler Research Group, we work on a broad array of projects that span analytical chemistry and chemical biology to neuroscience and bioengineering. We conduct research in bioanalytical chemistry and focus on the development of analytical methods for assaying complex microenvironments, including capillary electrophoresis separations, laser-based detectors, MALDI sampling techniques, and nanoliter volume NMR.
In addition to these information-rich approaches, new methodologies based on micro/nanofluidic sampling and single-cell isolations are used. Applying these techniques, we examine a range of neurochemical pathways in a variety of organisms. Studies include the distribution, metabolism, and dynamic release of neuropeptides and classical transmitters in a cell-specific manner. We are especially interested in the roles that such neuromodulatory compounds play in behavior, learning, and memory.
The development of novel toolsets enables new measurements that drive scientific advances. Just as important is the use of well-chosen animal models that cross the scale of simple to complex. Combining these two areas, we are advancing our understanding of new neurochemical pathways and brain function. You will find web pages on the techniques, the neurochemical pathways, and the models that we study.
Our Principal Investigator is Johnathon V. Sweedler.
Scroll down and check the tabs below or our website here to learn more.
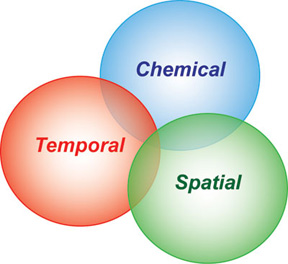 Our group works on a range of projects to improve analytical measurement technology, in most cases, pushing them to higher performance with smaller samples. The need to sample small volumes is obvious when working with single cells and subcellular domains. Thus, significant efforts use techniques with favorable scaling laws for low-volume samples. Capillary separations are well suited to working with small volume samples, as is mass spectrometry. Other approaches investigated include nanoliter volume NMR, microfluidics and microscopy. One goal is to develop approaches that achieve greater chemical information while obtaining either temporal or spatial information.
Our group works on a range of projects to improve analytical measurement technology, in most cases, pushing them to higher performance with smaller samples. The need to sample small volumes is obvious when working with single cells and subcellular domains. Thus, significant efforts use techniques with favorable scaling laws for low-volume samples. Capillary separations are well suited to working with small volume samples, as is mass spectrometry. Other approaches investigated include nanoliter volume NMR, microfluidics and microscopy. One goal is to develop approaches that achieve greater chemical information while obtaining either temporal or spatial information. Capillary electrophoresis (CE) is uniquely suited to the analysis of cellular samples due to the small inner diameter of typical capillaries (5-100 microns), which are well-matched to the size of isolated neurons (5-500 microns). The use of small capillaries leads to improved mass sensitivity and faster separations.
Capillary electrophoresis (CE) is uniquely suited to the analysis of cellular samples due to the small inner diameter of typical capillaries (5-100 microns), which are well-matched to the size of isolated neurons (5-500 microns). The use of small capillaries leads to improved mass sensitivity and faster separations.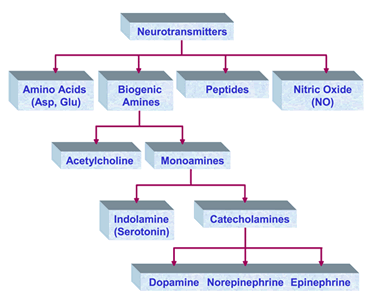 Cell to cell communication in the brain uses a diverse set of molecules, ranging from gaseous molecules such as nitric oxide (NO), classical transmitters such as glutamate, surprising molecules such as d-serine and a range of peptides. The Sweedler group studies these to understand how networks of neurons and associated supporting cells such as glia can work together to yield emergent properties that give rise to behaviors and memory. Specific questions address what molecules are present in specific cells and networks, how they change based on behavior or exposure to drugs, as well as their function.
Cell to cell communication in the brain uses a diverse set of molecules, ranging from gaseous molecules such as nitric oxide (NO), classical transmitters such as glutamate, surprising molecules such as d-serine and a range of peptides. The Sweedler group studies these to understand how networks of neurons and associated supporting cells such as glia can work together to yield emergent properties that give rise to behaviors and memory. Specific questions address what molecules are present in specific cells and networks, how they change based on behavior or exposure to drugs, as well as their function. 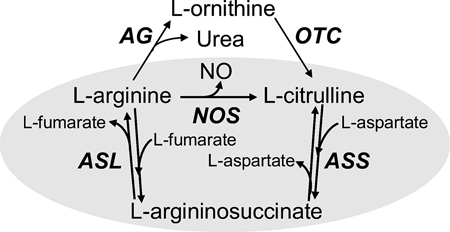 Nitric oxide (NO), a gaseous signal molecule, is involved in a variety of important biological functions in the cardiovascular system, the central and peripheral nervous system, the reproductive and the immune system. NO is typically generated through enzymatic conversion of arginine (Arg) to citrulline (Cit) by NO synthase (NOS). In addition to enzymatically produced NO, there may be another source of NO in neuronal tissues containing high levels of nitrite and ascorbate. Our group has been trying to understand these enzymatic and enzyme-independent NO production mechanisms chemically, temporally and spatially. Our model systems are functionally characterized and identified neurons of Aplysia californica, Pleurobranchaea californica, and Lymnaea stagnalis - three well-recognized models in cellular and system neuroscience. Capillary electrophoresis with laser-induced fluorescence detection (CE-LIF) is our method of choice, which is well-suited for the analysis of individual cells, primarily because of the high separation efficiencies, ultra-small-volume sample requirements, and sensitive detection methods associated with the technique.
Nitric oxide (NO), a gaseous signal molecule, is involved in a variety of important biological functions in the cardiovascular system, the central and peripheral nervous system, the reproductive and the immune system. NO is typically generated through enzymatic conversion of arginine (Arg) to citrulline (Cit) by NO synthase (NOS). In addition to enzymatically produced NO, there may be another source of NO in neuronal tissues containing high levels of nitrite and ascorbate. Our group has been trying to understand these enzymatic and enzyme-independent NO production mechanisms chemically, temporally and spatially. Our model systems are functionally characterized and identified neurons of Aplysia californica, Pleurobranchaea californica, and Lymnaea stagnalis - three well-recognized models in cellular and system neuroscience. Capillary electrophoresis with laser-induced fluorescence detection (CE-LIF) is our method of choice, which is well-suited for the analysis of individual cells, primarily because of the high separation efficiencies, ultra-small-volume sample requirements, and sensitive detection methods associated with the technique. Neuropeptidomics is a fairly recent term that indicates the global measurement and identification of the complement of peptides in a cell, tissue, or extracts from the brain or nervous system tissue. Obviously, peptide measurements have a much longer history, but the ability to approach global characterization and generation of lists of literally hundreds of peptides from a tissue represents a change in scale of such efforts.
Neuropeptidomics is a fairly recent term that indicates the global measurement and identification of the complement of peptides in a cell, tissue, or extracts from the brain or nervous system tissue. Obviously, peptide measurements have a much longer history, but the ability to approach global characterization and generation of lists of literally hundreds of peptides from a tissue represents a change in scale of such efforts. 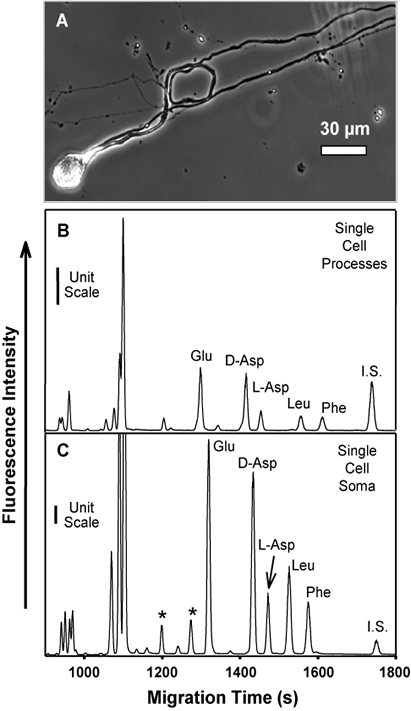
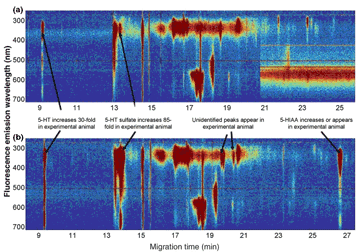
 What animal models do we use to study neuronal function? From "simpler" to "complex":
What animal models do we use to study neuronal function? From "simpler" to "complex":