Information about the Rhodes Laboratory.
 The Rhodes Laboratory is broadly interested in how genes and environment shape voluntary behavior. Most of our work involves performing behavioral experiments with mice and fish to answer basic questions about how lifestyle factors such as diet, drugs of abuse, social environment, and exercise affect the brain and behavior. Our approach combines behavioral, genetic, molecular, and histological techniques and involves extensive collaborations with different units on campus and outside campus including engineering, kinesiology, and nutrition.
The Rhodes Laboratory is broadly interested in how genes and environment shape voluntary behavior. Most of our work involves performing behavioral experiments with mice and fish to answer basic questions about how lifestyle factors such as diet, drugs of abuse, social environment, and exercise affect the brain and behavior. Our approach combines behavioral, genetic, molecular, and histological techniques and involves extensive collaborations with different units on campus and outside campus including engineering, kinesiology, and nutrition.
Our mice and fish are in two separate projects at the moment. Current mouse projects focus on uncovering origins of exercise-brain interactions, with two separate lines of research: one on muscle-brain communication and the other on brain activation associated with physical exertion. We also have an ongoing project exploring the impact of natural versus synthetic vitamin E on brain development. Meanwhile, our fish work currently has two main objectives which fill important gaps in our understanding of sex differences in the brain and the regulation of parental care. The first is uncovering the neuroendocrine mechanisms involved in the active feminization of the brain induced by social dominance and the latter on fathering behavior. The unique life history of the fish we are studying allows us to explore questions which are much more difficult if not impossible to study in mammals, but presumably, follow similar biological processes because of evolutionary ancestry.
Our Principal Investigator is Justin Rhodes.
Want to learn more about our lab? Click here.
 The Rhodes Lab houses the Mouse Cognition Core Facility for the Center for Nutrition, Learning, and Memory, a privately funded center which supports multiple projects exploring micronutrient impacts on cognitive performance in humans and animal models. The Core Facility was created to understand how nutrition can optimize brain health across the lifespan along with finding cognitive and behavioral paradigms that will elucidate the effect various nutritional compounds and supplements may have on the brain and behavior. The facility collaborates with both University of Illinois scientists as well as scientists from Abbott Nutrition. Additionally, our lab is also interested in how products of metabolism signal the brain to influence behavioral outcomes such as physical activity, cognitive performance, and food intake.
The Rhodes Lab houses the Mouse Cognition Core Facility for the Center for Nutrition, Learning, and Memory, a privately funded center which supports multiple projects exploring micronutrient impacts on cognitive performance in humans and animal models. The Core Facility was created to understand how nutrition can optimize brain health across the lifespan along with finding cognitive and behavioral paradigms that will elucidate the effect various nutritional compounds and supplements may have on the brain and behavior. The facility collaborates with both University of Illinois scientists as well as scientists from Abbott Nutrition. Additionally, our lab is also interested in how products of metabolism signal the brain to influence behavioral outcomes such as physical activity, cognitive performance, and food intake. It is now established that incorporating regular exercise into the life routine is crucial for maintaining cognitive health throughout the lifespan, however, the mechanisms for the pro-cognitive effects of exercise are not well understood. Elucidating and unequivocally establishing these mechanisms holds the key to discover novel and more efficient ways to maintain, promote and improve cognitive performance, perhaps even in the absence of physical activity. We and many others have documented numerous long-term changes in the central nervous system (CNS) from exercise training which likely support enhanced cognition. However, the acute effects of exercise which ultimately must cause the long-term CNS adaptations are not known. Moreover, it is not clear where the acute signals come from, the periphery (e.g., muscles, blood pressure) or within the brain itself. We believe it is crucial to identify those specific acute events associated with exercise or physical exertion that when repeated contribute to the chronic neurological changes that have been so well documented and presumably related to the cognitive enhancement. This is because if we seek to replicate exercise’s pro-cognitive influence, then it likely will be necessary to recapitulate the acute events and repeat them to cause the desired long-term cognitive benefits, as occurs with exercise training.
It is now established that incorporating regular exercise into the life routine is crucial for maintaining cognitive health throughout the lifespan, however, the mechanisms for the pro-cognitive effects of exercise are not well understood. Elucidating and unequivocally establishing these mechanisms holds the key to discover novel and more efficient ways to maintain, promote and improve cognitive performance, perhaps even in the absence of physical activity. We and many others have documented numerous long-term changes in the central nervous system (CNS) from exercise training which likely support enhanced cognition. However, the acute effects of exercise which ultimately must cause the long-term CNS adaptations are not known. Moreover, it is not clear where the acute signals come from, the periphery (e.g., muscles, blood pressure) or within the brain itself. We believe it is crucial to identify those specific acute events associated with exercise or physical exertion that when repeated contribute to the chronic neurological changes that have been so well documented and presumably related to the cognitive enhancement. This is because if we seek to replicate exercise’s pro-cognitive influence, then it likely will be necessary to recapitulate the acute events and repeat them to cause the desired long-term cognitive benefits, as occurs with exercise training. Clownfishes typically live most of their life on or in very close proximity to a sea anemone in small groups from 3 up to 6 or 7 depending on the size of the anemone. In these groups, the largest is usually the reproductive female, the second-largest, the male and the smaller individuals are undifferentiated. If the female is removed, the male will transform into a female in a matter of weeks. Immediately, within the first few minutes, the male will begin to display female-typical behavior, e.g., court the other smaller fish. Within a few weeks, the testes of the male will have been absorbed, and ovaries will replace them. In addition, the size of the fish will increase, and the genitalia will change shape to allow for the passage of eggs instead of sperm through the genital duct. At the same time, the largest of the undifferentiated fish will transform into a male following a similar time course.
Clownfishes typically live most of their life on or in very close proximity to a sea anemone in small groups from 3 up to 6 or 7 depending on the size of the anemone. In these groups, the largest is usually the reproductive female, the second-largest, the male and the smaller individuals are undifferentiated. If the female is removed, the male will transform into a female in a matter of weeks. Immediately, within the first few minutes, the male will begin to display female-typical behavior, e.g., court the other smaller fish. Within a few weeks, the testes of the male will have been absorbed, and ovaries will replace them. In addition, the size of the fish will increase, and the genitalia will change shape to allow for the passage of eggs instead of sperm through the genital duct. At the same time, the largest of the undifferentiated fish will transform into a male following a similar time course. Despite decades of intense research since the discovery that new neurons are continuously generated in the adult mammalian brain, their function remains a mystery. Determining the role of neurogenesis in healthy and diseased brains has broad implications for our understanding of learning, memory, behavior, and regenerative medicine. We are taking advantage of recent advances in optogenetic technology to design tools for studying new neurons in vivo. By combining optogenetics (which uses light to control genetically modified cells) with our lab’s expertise in behavior, we are making progress towards solving one of the biggest mysteries in neuroscience.
Despite decades of intense research since the discovery that new neurons are continuously generated in the adult mammalian brain, their function remains a mystery. Determining the role of neurogenesis in healthy and diseased brains has broad implications for our understanding of learning, memory, behavior, and regenerative medicine. We are taking advantage of recent advances in optogenetic technology to design tools for studying new neurons in vivo. By combining optogenetics (which uses light to control genetically modified cells) with our lab’s expertise in behavior, we are making progress towards solving one of the biggest mysteries in neuroscience.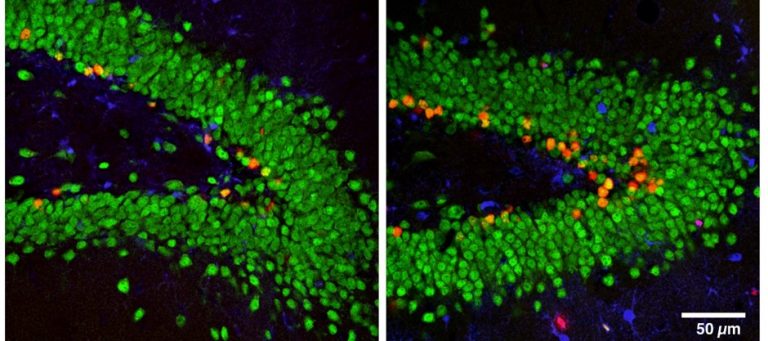 A large literature has established the critical importance of aerobic exercise for maintaining physical and mental health throughout the lifespan, yet average daily levels of physical activity continue to decline in western society. The major obstacle preventing the global therapeutic application of exercise is that for many people, the desire to exercise is low, and appears to be declining. Moreover, it is not clear how to increase motivation for exercise. Physical exercise can be rewarding and addictive in certain individuals, suggesting that motivation for physical activity can be increased. However, relatively few studies have investigated the neurobiology of increased motivation for exercise and our understanding of how to neurologically increase the desire to exercise is rudimentary at best.
A large literature has established the critical importance of aerobic exercise for maintaining physical and mental health throughout the lifespan, yet average daily levels of physical activity continue to decline in western society. The major obstacle preventing the global therapeutic application of exercise is that for many people, the desire to exercise is low, and appears to be declining. Moreover, it is not clear how to increase motivation for exercise. Physical exercise can be rewarding and addictive in certain individuals, suggesting that motivation for physical activity can be increased. However, relatively few studies have investigated the neurobiology of increased motivation for exercise and our understanding of how to neurologically increase the desire to exercise is rudimentary at best. We have developed a method, using sophisticated video tracking software from CleverSysems Inc., to measure physical activity in home cages 24 hours a day 7 days a week in hundreds of mice each generation. We are currently breeding lines of mice for high levels of cage activity while maintaining other un-selected lines to serve as controls. The goal is to identify how genes influence behavior at multiple levels of biological organization from the genes to the development and function of a nervous system.
We have developed a method, using sophisticated video tracking software from CleverSysems Inc., to measure physical activity in home cages 24 hours a day 7 days a week in hundreds of mice each generation. We are currently breeding lines of mice for high levels of cage activity while maintaining other un-selected lines to serve as controls. The goal is to identify how genes influence behavior at multiple levels of biological organization from the genes to the development and function of a nervous system.