 Over the past decade, a new scientific discipline has emerged, integrating mechanics with biology to create complex engineered living systems. The building blocks, different cell types in an instructive environment, can be assembled in various ways to promote the emergence (or natural evolution and interaction) of the cells in a system with well-defined functionality. These functions could include sensing, information processing, protein expression, and actuation.
Over the past decade, a new scientific discipline has emerged, integrating mechanics with biology to create complex engineered living systems. The building blocks, different cell types in an instructive environment, can be assembled in various ways to promote the emergence (or natural evolution and interaction) of the cells in a system with well-defined functionality. These functions could include sensing, information processing, protein expression, and actuation.
Scientists at the forefront of this research envision the extent of cellular and biological systems to include devices that are able to sense their surroundings and perform accordingly. Examples of cellular systems that can have real-world implications in medicine and the environment include:
- a biological “robot” or organism that has the ability to recognize a chemical gradient and produce a response, such as moving towards an oil spill for environmental remediation or neutralizing a dangerous toxin within the body;
- an engineered “organ” to sense drug and protein levels or glucose concentration in the bloodstream and send the information to a feedback loop or insulin pump;
- a micro-fluidic system acting as a human organ platform (heart, lung, or liver to eliminate harmful and costly drug development and testing; and
- plants with neurons that can send out a signal for water or nutrients after sensing a local deficiency.
All of these – and countless more – could potentially be achieved with the use of a few cell types, specifically neurons, endothelial cells, and muscle cells, which can be differentiated from stem cells.
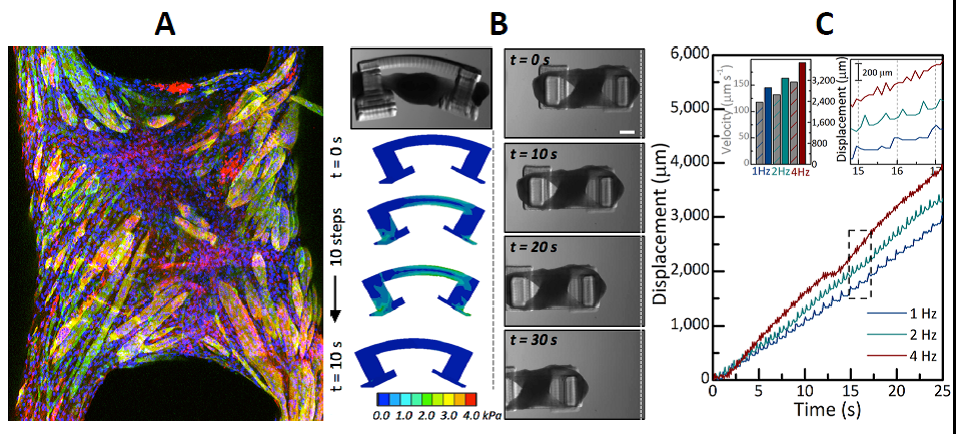
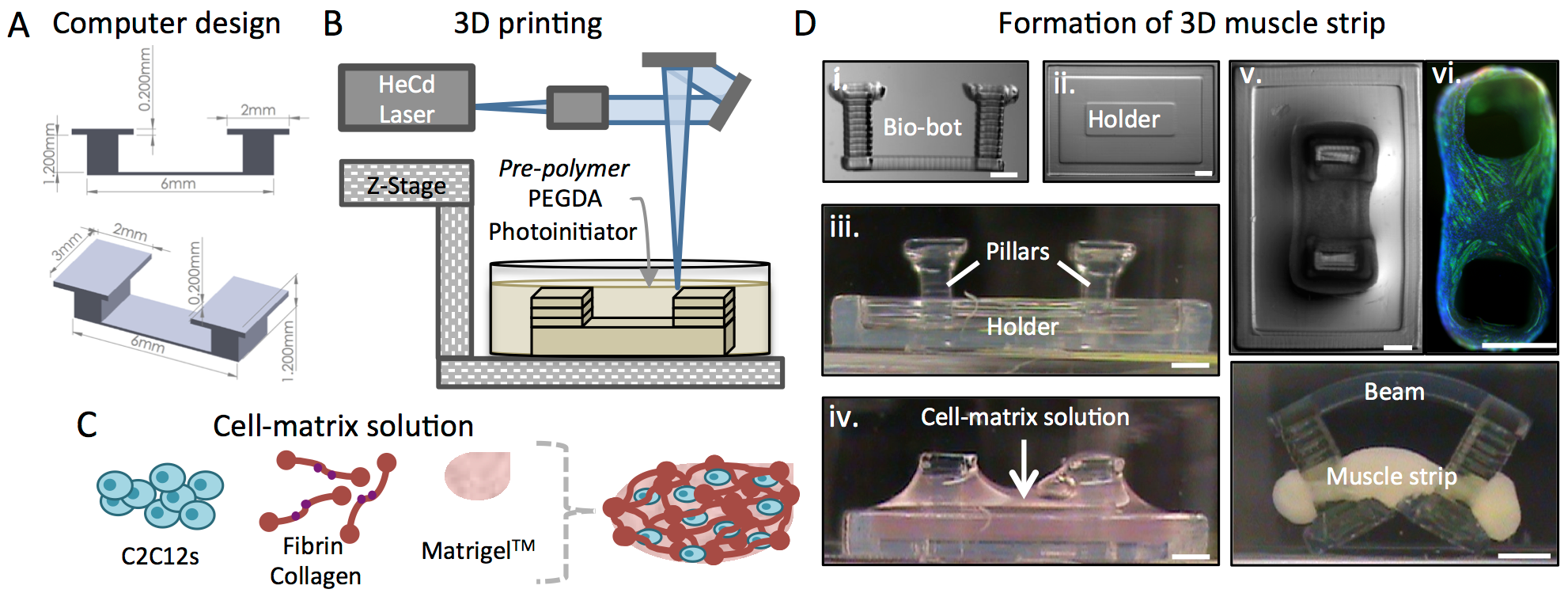
Cell-based soft robotic devices could have a transformative impact on our ability to design machines and systems that can dynamically sense and respond to a range of complex environmental signals. We demonstrate an interdisciplinary and novel integration of biomaterials, tissue engineering, and 3D printing to forward engineer controllable centimeter-scale biological machines capable of locomotion. Due in part to their elastic nature and the living components that can permit a dynamic response to environmental and applied stimuli, these biological machines can have diverse applications and represent a significant advancement towards high-level functional control over soft bio-robotic systems.
Combining biological components such as cells and tissues with soft robotics can enable the fabrication of biological machines with the ability to sense, process signals, and produce force. An intuitive demonstration of a biological machine is one that can produce motion in response to controllable external signaling. While cardiac cell-driven biological actuators have been demonstrated, the requirements of these machines to respond to stimuli and exhibit controlled movement merit the use of skeletal muscle, the primary generator of actuation in animals, as a contractile power source. We are interested in developing 3D printed hydrogel ‘bio-bots’ powered by the actuation of engineered mammalian skeletal muscle to result in net locomotion of the bio-bot.
The “biological parts” comprising many higher-level systems have been assembled to date as individual machine components with one or two cell types. Muscle cells have been used for biological actuators and neurons or neuronal networks assemble into sensory clusters or processors. The challenge, then, is to coordinate the cell types in a way that allows for new functionalities and applications in various fields. A complete cellular system needs many different components in order to be self-sustaining and autonomous: muscle cells that allow for movement, neuromuscular junctions to interface between muscle and neurons for control and processing, a vasculature supported by endothelial cells to provide nutrients and oxygen, and an exoskeleton for long-term viability and protection.
 Laboratory of Integrated Biomedical Micro/Nanotechnology and Applications’ (LIBNA) vision in integrating biology and medicine with micro and nanotechnology can be categorized into two broad areas, namely how micro/nano-fabrication can help solve problems in life sciences (such as diagnostics, therapeutics, and tissue engineering), and how we can learn more from life science to solve important problems in micro/nano-science and engineering (such as bio-inspired self-assembly, etc.).
Laboratory of Integrated Biomedical Micro/Nanotechnology and Applications’ (LIBNA) vision in integrating biology and medicine with micro and nanotechnology can be categorized into two broad areas, namely how micro/nano-fabrication can help solve problems in life sciences (such as diagnostics, therapeutics, and tissue engineering), and how we can learn more from life science to solve important problems in micro/nano-science and engineering (such as bio-inspired self-assembly, etc.). 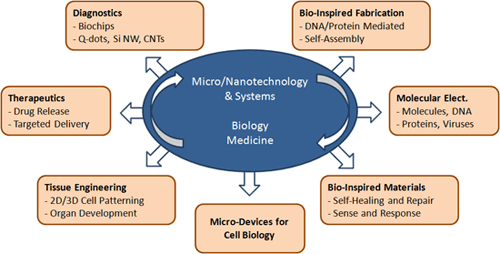 Thrust areas of research in our group are:
Thrust areas of research in our group are:
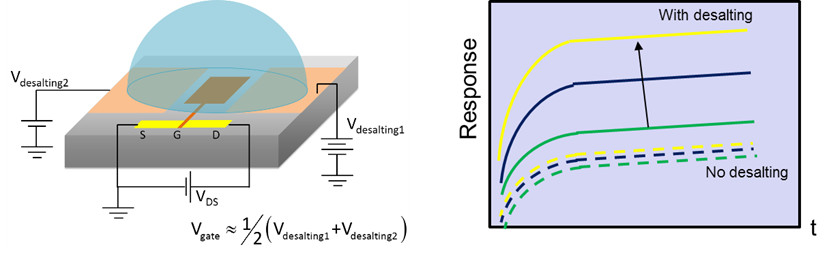


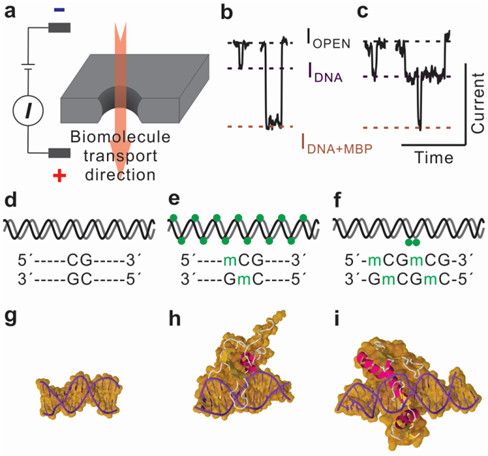
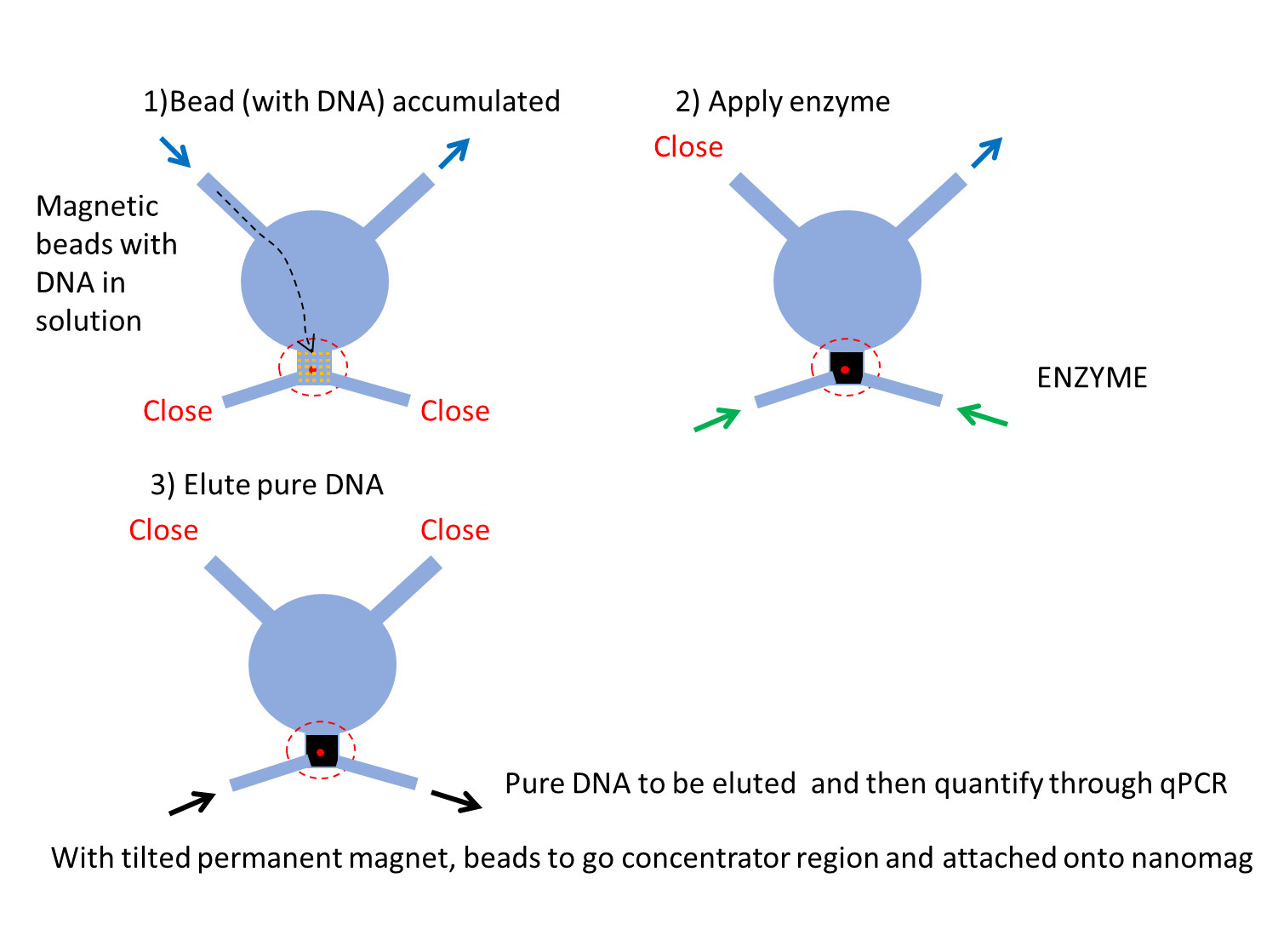
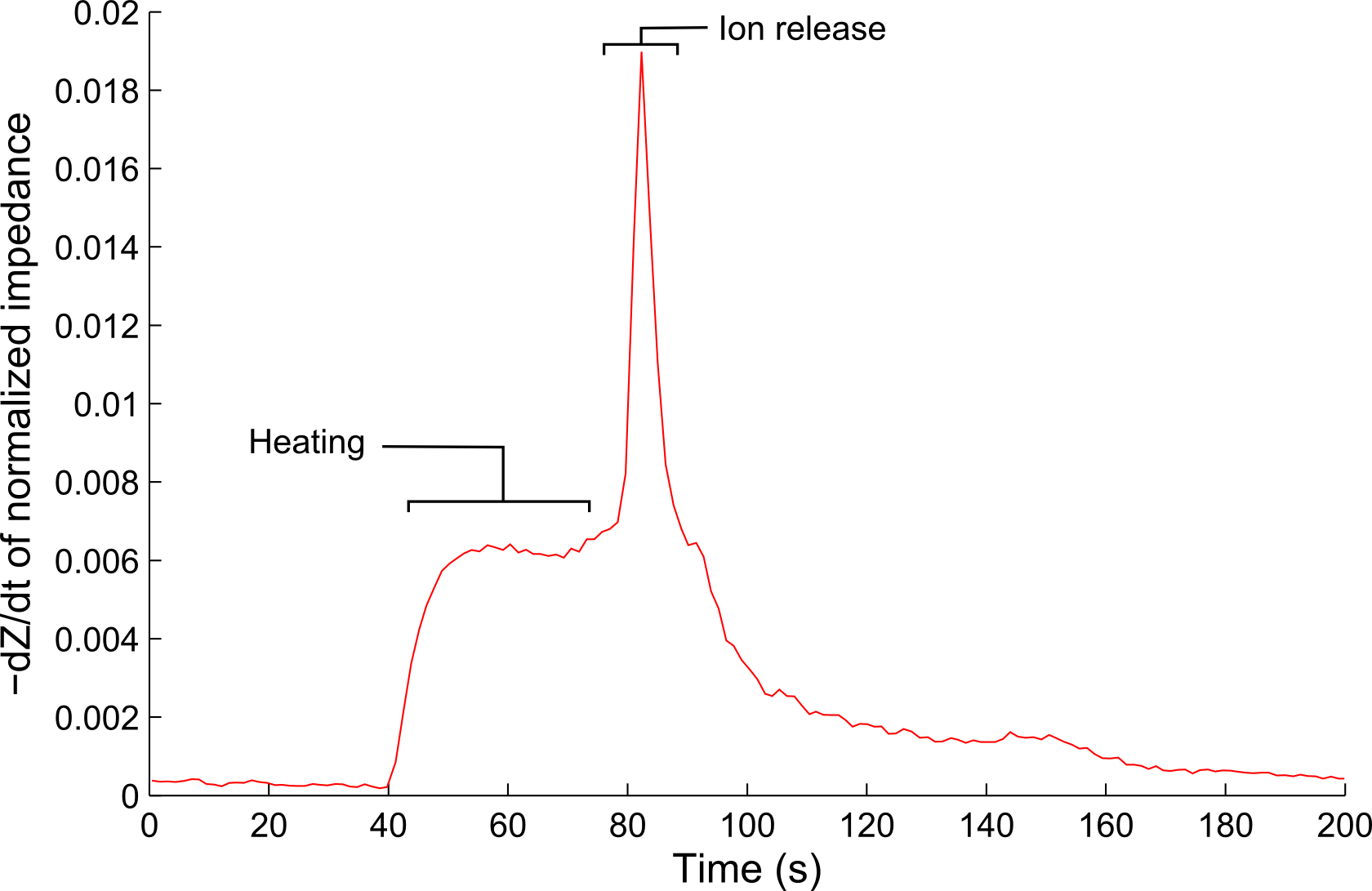
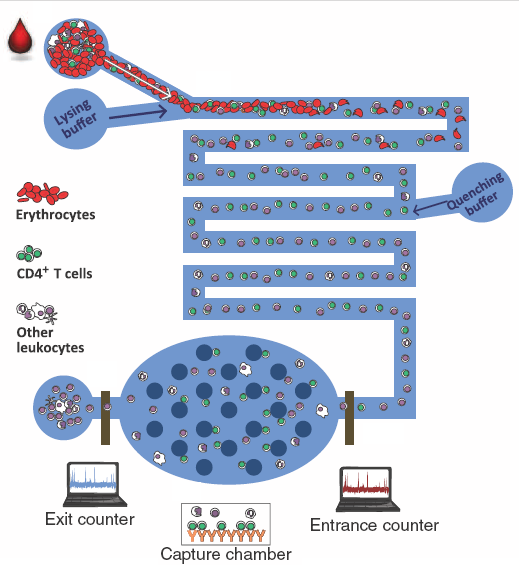
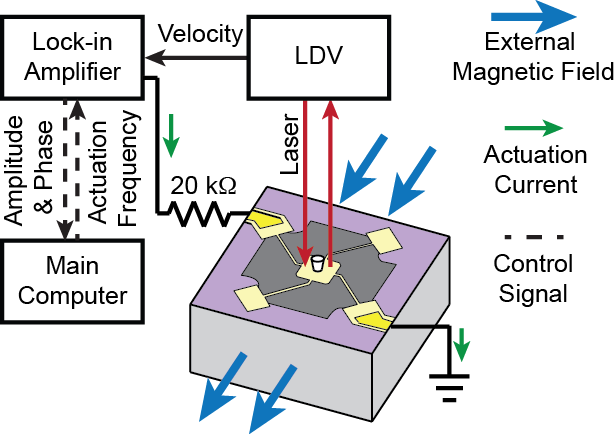 A complex relationship exists between the behavior of a cell and its physical properties. There is a strong interest in understanding the mechanisms through which the physical properties of a cell influence growth, differentiation, cell cycle progression, and apoptosis. Nanobiomechanics, which is the measuring of material properties at the smallest length scale, is an emerging area of research that has the ability to evolve the study of human disease. This field has helped elucidate mechanisms of disease progression and provided additional tools in the fight against the disease.
A complex relationship exists between the behavior of a cell and its physical properties. There is a strong interest in understanding the mechanisms through which the physical properties of a cell influence growth, differentiation, cell cycle progression, and apoptosis. Nanobiomechanics, which is the measuring of material properties at the smallest length scale, is an emerging area of research that has the ability to evolve the study of human disease. This field has helped elucidate mechanisms of disease progression and provided additional tools in the fight against the disease. Over the past decade, a new scientific discipline has emerged, integrating mechanics with biology to create complex engineered living systems. The building blocks, different cell types in an instructive environment, can be assembled in various ways to promote the emergence (or natural evolution and interaction) of the cells in a system with well-defined functionality. These functions could include sensing, information processing, protein expression, and actuation.
Over the past decade, a new scientific discipline has emerged, integrating mechanics with biology to create complex engineered living systems. The building blocks, different cell types in an instructive environment, can be assembled in various ways to promote the emergence (or natural evolution and interaction) of the cells in a system with well-defined functionality. These functions could include sensing, information processing, protein expression, and actuation.