Bonthuis Lab - Research Interest

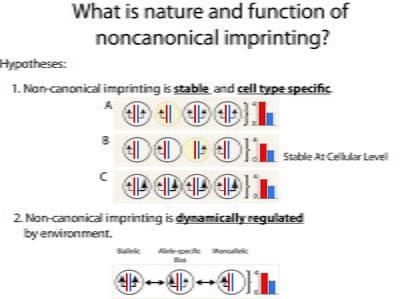
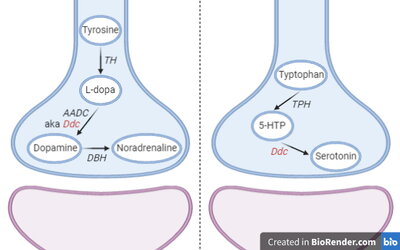
My research objective is to understand how genes, epigenetic regulation of gene expression, and hormone signaling in the brain interact to regulate social behaviors. Most recently, my focus has been to investigate noncanonical genomic imprinting in the monoamine neurotransmitter system of the brain and the functional consequences on mammalian behavior. Genomic imprinting is an epigenetic phenomenon in mammals where the expression of an allelic copy of a gene depends on whether it was inherited from the mother or the father. A major theory about genomic imprinting in the brain is that it functions to regulate social behaviors between mothers and offspring, and even between adult social groups. Using newly developed allelic-reporter mice made by CRISPR mediated knock-in mutagenesis, and neural histology methods, I have begun to identify which monoaminergic neurons in the brain are subject to genomic imprinting effects. In addition, genetic mouse models and ongoing behavioral experiments are characterizing which aspects of social behavior are impacted by genomic imprinting in the monoamine neurotransmitter system. Moving forward, my research will use genomic imprinting effects as a marker to identify cell-types and circuits important for regulating complex social behaviors in mammals.
Our Principal Investigator is Paul Bonthuis.
Check out our website here.